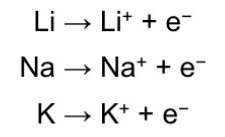Learning Outcomes
At the end of this section, you should be able to:
- Describe the physical and chemical properties of Group I elements.
- Predict the properties of Group I elements using the Periodic Table.
What have you observed from the videos?
Write your answers on your worksheet.
Write your answers on your worksheet.
About Group I
Group I elements:
- Can react with water to form alkalis.
- Are also known as alkali metals.
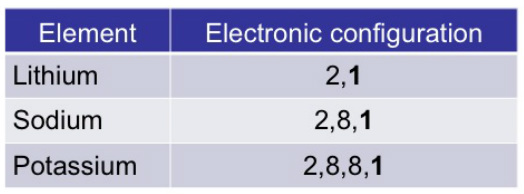
- Contain one electron each in their valence shells
1. Physical Properties
|
Group I elements:
|
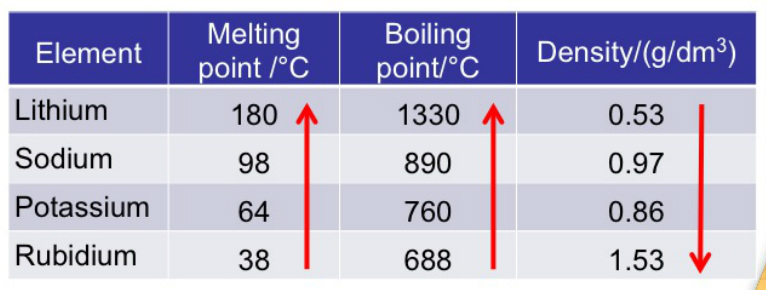
Trends in Physical Properties
|
Melting and boiling points decrease down the group.
|
Densities increase down the group.
|
2. Chemical Properties
Alkali metals have one electron in its valence shell.
Hence, they have similar chemical properties.
Alkali metals are very reactive.
Alkali metals react with cold water to form alkali and hydrogen gas.
Hence, they have similar chemical properties.
Alkali metals are very reactive.
- E.g. sodium tarnishes easily in air to form an oxide.
- It is stored in oil to prevent reaction with air and moisture.
Alkali metals react with cold water to form alkali and hydrogen gas.
- alkali metal + water → alkali + hydrogen gas
Trends in Chemical Properties
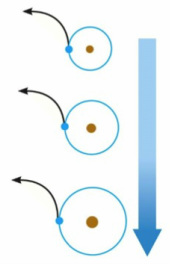
Each alkali metal loses its valence electron easily to attain a noble gas structure.
A positive ion is formed.
A positive ion is formed.
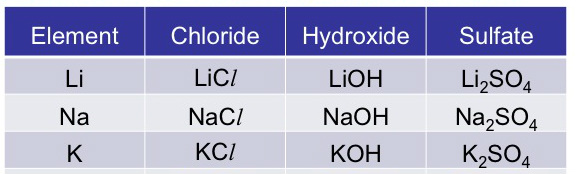
Compounds of Alkali metals are:
- Ionic
- Soluble in water
- Similar chemical formulae